RNDr. Rostislav Škrabana, PhD
 |
 |
Structure means a mode of existence, which determines the function. Laboratory of Structural Proteomics studies the structural and biophysical transformation of proteins involved in neurodegenerative processes and structural principles of antibody effects on disease-modified proteins. Our focus is aimed at proteins with a high degree of structural disorder, so-called intrinsically disordered proteins (IDPs), which are often involved in devastating human illnesses, namely tau protein (Alzheimer’s disease), synuclein (Parkinson disease) and prion protein (spongiform encephalopathy).
We employ structural biology tools (X-ray crystallography) corroborated with dynamic biophysical methods of protein structure analysis (surface plasmon resonance, light scattering, molecular spectroscopy, mass spectrometry) and supported by molecular modeling in silico.
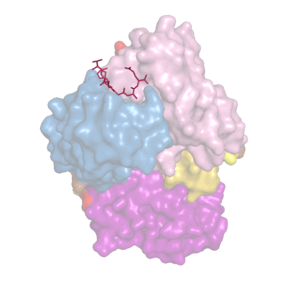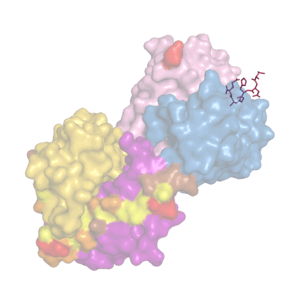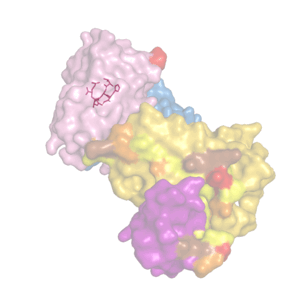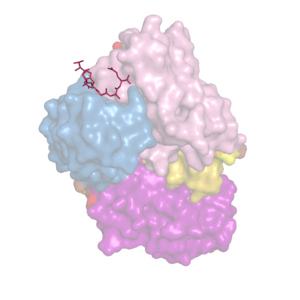
We analyze recombinant forms of tau proteins and their interactions with structure-specific monoclonal antibodies.
Lab Members
- Ing. Ondrej Cehlár, PhD
- MSc. Branislav Kováčech, PhD
- MSc. Klaudia Mešková (PhD student)
- MSc. Stefana Njemoga (PhD student)
- MSc. Katarína Tomková (research assistant)
Awards
- Prize of the Czech Society for Structural Biology for an outstanding student poster
- Stefana Njemoga, Institute of Neuroimmunology, Slovak Academy of Sciences received the prize for her poster at the XVIII Discussions in Structural Molecular Biology and the 5th User Meeting of CIISB that was held in Nove Hrady, South Bohemia, on 24-26 March 2022.
- Poster title: Multiapproach docking study for binding of intrinsically disordered tau peptides to monoclonal antibodies
Projects – funding
| Non-globular proteins in the era of Machine Learning (ML4IDP) |
| COST Action CA21160 |
| Duration: 11/2022-10/2026 |
| ………… |
| Conformational properties of intrinsically disordered protein tau with the emphasis on the C-terminus of its molecule |
| VEGA 2/0145/19 |
| Duration: 01/2019 – 12/2022 |
| ………… |
| Non-globular proteins – from sequence to structure, function and application in molecular physiopathology (NGP-NET) |
| COST Action BM1405 |
| Duration: 05/2015-03/2019 |
| ………… |
| Structural study of multiple homologous epitopes present on the molecule of the intrinsically disordered protein tau. |
| VEGA 02/0177/15 |
| Duration: 01/2015-12/2018 |
| ………… |
| Biophysics and structure of intrinsically disordered polypeptides studied on the N-terminal tail of tau protein: implications for neurodegenerative tauopathies. |
| VEGA 02/0163/13 |
| Duration: 01/2013-12/2016 |