Structural Biology and Biophysics symposium
Structural Biology and Biophysics symposium, will be organized on 30th. of april 2026, in the conference room of the Institute of virology, Dubravska cesta 9.
Invitation to Structural Biology and Biolophysics Symposium
Poster presentation:
Students have the opportunity to present a poster. Please send the abstract of your poster presentation by email to: ondrej.cehlar@savba.sk
Abstract of the invited lecture: Understanding the versatility of S1–P1 nucleases
Tomáš Kovaľ tomas.koval@ibt.cas.cz
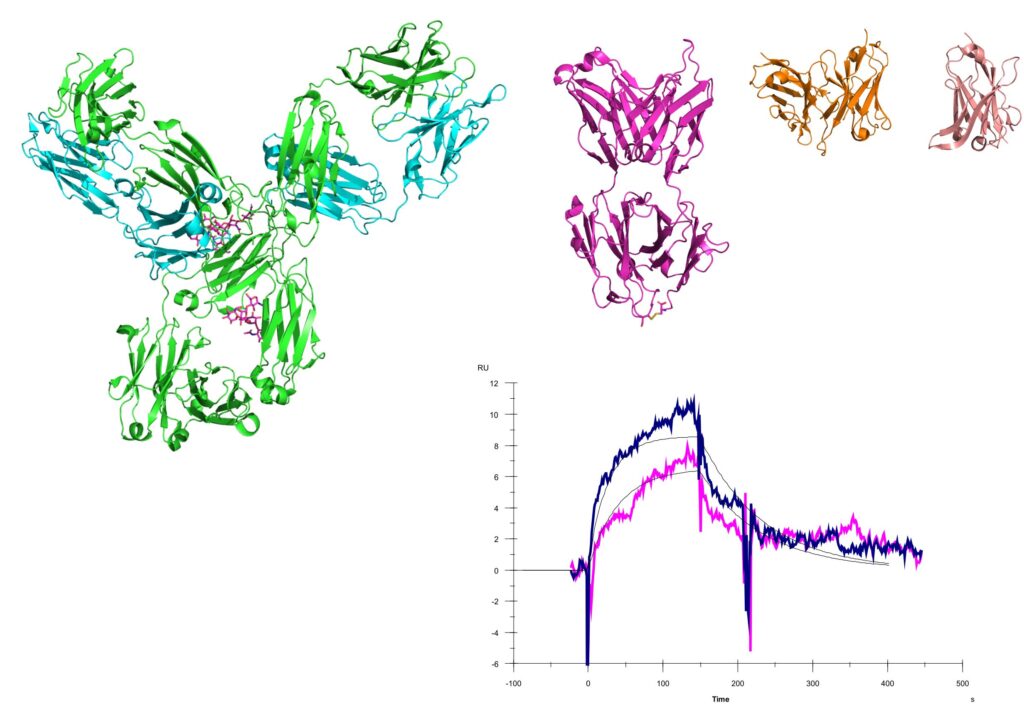
S1–P1 nucleases are medium-sized enzymes (25–40 kDa) that share a distinctive active site built around a cluster of three zinc ions. They function as phosphoesterases, cleaving the P‑O3’ bond in nucleic acids and producing 5′‑mononucleotides. What makes this enzyme family particularly interesting is its broad substrate range: S1–P1 nucleases can cleave almost any nucleic acid, often without regard to sequence or secondary structure. Members of this family are found in plants, fungi, trypanosomatids, and some bacteria, where they contribute to nutrient scavenging, programmed cell death, and even pathogen–host interactions. Several have even found applications in biochemistry and biotechnology (e.g. S1 nuclease protection assay) [1].
In our work, we focus on two representative enzymes: S1 nuclease from Aspergillus oryzae and SmNuc1 from the opportunistic pathogen Stenotrophomonas maltophilia. Both proteins form crystals that diffract to subatomic resolution and can accommodate any naturally occurring nucleotide—or even nucleotide pairs [2]. These high‑resolution structures allow us to investigate subtle structural features and clarify what drives the remarkable substrate promiscuity of the S1–P1 family. At the same time, they help explain why different family members display distinct preferences, such as acidic vs. basic pH optima or differing activities toward DNA and RNA, providing new insight into both their biological roles and their potential biotechnological applications.
[1] T. Koval, J. Dohnalek Biotechnol. Adv. (2017) 10.1016/j.biotechadv.2017.12.007.
[2] K. Adámková, M. et al. FEBS J., 292, (2025) doi/10.1111/febs.17265